Your Resource Hub for Justice and Empowerment
Access free tools, videos and resources to help you understand your options and take action.
A Central Resource for Knowledge, Action, and Clarity
Curated by Attorney Eric Esteban Cardenas
I want this Resource Hub to be your go-to destination for tools, insights, and guidance to help you tackle tough legal issues. Whether you need answers about your legal rights, want to explore detailed case investigation "Deep Dives", or use our innovative Case Qualifying Quiz to see if you have a case instantly, this hub is here for you.
With blog articles, step-by-step guides, and social media content from our Facebook, Instagram, YouTube, and TikTok, the Resource Hub give you the knowledge to take control of your legal journey.
Knowledge is power. Use these tools to take the first step toward rebuilding your life. I'll keep expanding this hub, so if there's a topic you'd like covered, please let me know. Enjoy. - Eric
The Blog
Deep Dives, Investigations, Step-By-Step Guides, and Articles.
Bard PowerPort Lawsuit 2024: Filing Claims for Device Malfunctions and Injuries
Bard PowerPort Lawsuit 2024 | A Deep Dive into Filing Claims for Device-Related Injuries
“When a catheter fractures, broken pieces may circulate in the bloodstream, potentially causing severe internal damage. Emergency surgery is often required to retrieve the fractured pieces and repair the affected areas.”
Are You Eligible for a Bard Powerport Lawsuit?
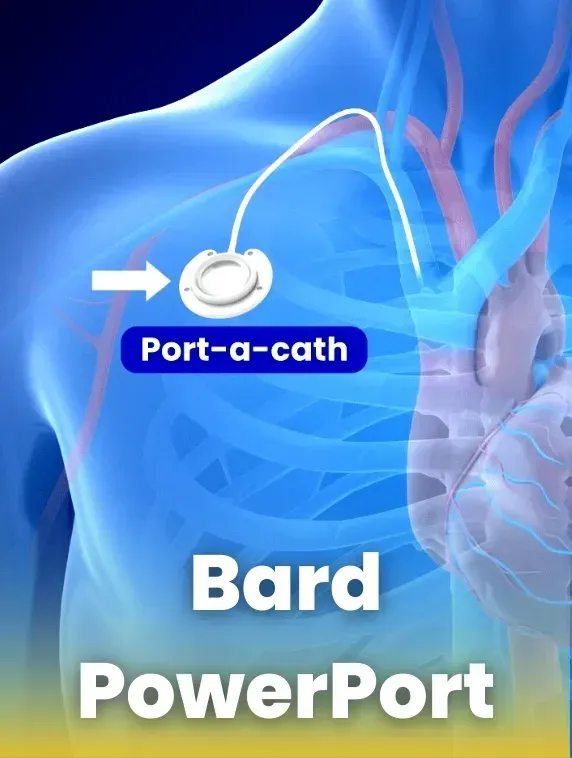
Bard PowerPort devices, also known as port-a-caths, are totally implantable venous access devices (TIVADs) designed to provide repeated access to the vascular system for delivering medications. While these devices are widely used in medical treatments, lawsuits claim they may pose significant risks to patients due to design defects.
What Are the Problems with Bard PowerPort?
Four primary issues have been identified with Bard PowerPort devices:
Fracture: The catheter tubing is prone to cracking and breaking. This can lead to serious complications such as blood clots, internal bleeding, vein or tissue damage, and migration of the catheter into the heart or lungs. In severe cases, surgical intervention is required to remove the device.
Infection: Catheter-related bloodstream infections are among the most deadly complications. Bacteria can lodge in fractures, forming biofilms that make infections resistant to treatment. These infections are a leading cause of death in hospitals.
Thrombosis: Blood clots can form around the catheter, leading to reduced blood flow or further complications. This is a weaker basis for cases compared to fractures or infections.
Detachment: In rare cases, the device can become dislodged from its implantation site. Detached catheters may cause damage as they move through the bloodstream.
Why Do These Issues Happen?
The failures are often linked to the materials used in the PowerPort device. Barium sulfate particles in the catheter can corrode over time, weakening the tubing and leading to fractures. Despite the availability of safer alternatives, such as encapsulated barium sulfate or surface-modifying additives (SMAs) to prevent degradation, these improvements have not been implemented consistently.
Holding Manufacturers Accountable
Our law firm is committed to helping individuals harmed by potentially defective medical devices. We are not afraid to take on large corporations like Bard, which holds a significant share of the vascular device market, to seek justice for those affected.
If you or a loved one has experienced complications such as fractures, infections, or other injuries due to a Bard PowerPort device, contact us today for a free consultation. Together, we can pursue accountability and compensation for the harm caused.
Updates on Bard PowerPort Lawsuits
Some Bard PowerPort devices have been linked to defects that may lead to serious injuries or severe infections in patients.
Numerous lawsuits have been filed against the manufacturer, Bard, and these cases have been consolidated into multidistrict litigation (MDL) in the U.S. District Court for the District of Arizona.
MDL 3081, which addresses the Bard PowerPort lawsuits, is presided over by Judge David G. Campbell.
Multidistrict litigation is a federal legal process designed to streamline complex cases involving similar claims and injuries. Unlike a class action lawsuit, where plaintiffs are treated as a single group, MDL allows individual cases to retain their unique circumstances while benefiting from a coordinated legal process.
Understanding the Bard PowerPort Device
The Bard PowerPort is an implanted medical device designed to provide easy access to a patient’s bloodstream for intravenous therapy or medication injections. Manufactured by Bard Access Systems Inc., a subsidiary of Becton, Dickinson and Company, the PowerPort is marketed as being capable of withstanding higher injection pressures than standard ports, making it suitable for rapid injection of fluids.
Key Components of the Bard PowerPort Device:
The Injection Port: Implanted under the skin, typically in the chest or arm, this port provides a site for needle insertion. It is made of a self-sealing material, allowing for multiple needle punctures while maintaining durability.
The Polyurethane Catheter: A flexible tube connected to the injection port, this catheter is inserted into a large central vein that carries blood to the heart. It facilitates the delivery of fluids and medications directly into the bloodstream.
To use the PowerPort, medical professionals employ a specialized needle called the PowerLoc needle. This needle accesses the bloodstream through the injection port, enabling the administration of medications and the collection of blood samples.
Materials Used in the Bard PowerPort Device
Bard PowerPort catheters are made from a polyurethane material called Chronoflex AL, which combines polyurethane and barium sulfate. While barium sulfate is included to make the device visible on X-rays, it may contribute to serious risks:
Fracturing: Barium sulfate particles can break away from the catheter, weakening its structure and making it prone to cracking.
Migration: Once fractured, catheter pieces can move to other parts of the body, potentially causing damage to tissues, veins, or organs.Exposure to content that normalizes disordered eating, self-harm, and unrealistic body standards.
These material defects have been cited as a significant factor in many lawsuits filed against Bard PowerPort, alleging that the design and materials make the device unsafe for patients.
Serious Risks Linked to Bard PowerPort Devices
The Bard PowerPort Lawsuit alleges that defects in certain Bard PowerPort catheters can lead to severe injuries and life-threatening complications. These issues stem from the susceptibility of the catheter's components to fracturing and migration, which can cause significant harm to patients.
Common Injuries and Complications Linked to the Bard PowerPort Device:
Catheter fractures
Catheter migration
Deep vein thrombosis (DVT)
Vein and blood vessel damage
Catheter infections and necrosis
Blood clots
Arterial punctures
Heart attacks (myocardial infarction)
Hematomas or hemorrhages
Hemothorax
Pulmonary pseudoaneurysms
Pulmonary embolism
Tachycardia
Collapsed lungs
Stroke
Other serious injuries
What Happens When a Catheter Fractures?
When a catheter fractures, broken pieces may circulate in the bloodstream, potentially causing severe internal damage. In many cases, emergency surgery is required to retrieve the fractured pieces and repair the affected areas.
Injuries Caused by Catheter Fractures:
Cardiac arrhythmia
Pulmonary embolism
Cardiac punctures
Blood clots
Hematomas
Lacerations to blood vessels
Risks of Infections from Bard PowerPort Devices
Catheter infections can introduce harmful pathogens into the body, leading to severe complications. These infections may become resistant to treatment and, if left unchecked, can result in life-threatening conditions.
Injuries Caused by Catheter-Related Infections:
Bloodstream infections
Swelling, tenderness, and soreness around the PowerPort site
Tissue necrosis near the PowerPort
These complications underline the serious risks posed by Bard PowerPort devices and support the claims made in ongoing lawsuits. If you or a loved one have experienced any of these injuries, it’s important to seek legal and medical assistance immediately.
Steps to Determine Your Eligibility for a Bard PowerPort Lawsuit
If you or a loved one suffered injuries after using a Bard implantable port catheter device, you may qualify to file a lawsuit against the manufacturer. Contact Eric Cardenas LAW, PLLC for a free consultation, or use the Instant Qualifying Quiz on this page to check your eligibility today.
Our firm has experience litigating defective medical device cases and holding manufacturers accountable for their negligence.
How to Address Bard PowerPort Injuries Promptly
If you suspect a problem with your PowerPort device or experience pain near the implantation site, seek immediate medical attention. Early treatment is critical to prevent further harm.
Follow all medical advice and take steps to minimize the long-term impact of your injuries, including financial costs. Prompt action can strengthen your legal case.
Why You Need a Bard PowerPort Injury Lawyer
The civil litigation process can be complex and time-consuming, especially in cases involving defective medical devices. An experienced attorney can help you:
Determine liability for the device's defects.
Gather evidence to support your case.
Assess damages, both economic and non-economic.
Build a strong legal argument against the manufacturer.
Navigate the litigation process to ensure you receive fair compensation.
Contact Eric Cardenas LAW today for a free, no-obligation consultation. Keep in mind that the statute of limitations restricts how long you have to file a claim after an injury, so don’t delay in seeking legal advice.
What Evidence Strengthens a Bard PowerPort Lawsuit?
Evidence is critical in a personal injury & mass tort case, particularly when dealing with defective medical devices. While your lawyer will assist with collecting evidence, you should begin preserving relevant materials as soon as possible.
Examples of Key Evidence for Bard PowerPort Lawsuits:
Medical records documenting your treatment and injuries.
Medical bills and other receipts related to costs.
Employment records showing lost wages or reduced earning capacity.
Treatment information from healthcare providers.
Photos and videos of injury progression.
Written or video personal accounts describing your experience.
Witness testimony, including statements from family or friends.
How Damages Are Calculated in Bard PowerPort Cases
Damages represent the losses caused by your injury, including economic and non-economic harm.
Types of Compensation Available in Bard PowerPort Lawsuits:
Medical expenses, including hospital stays, surgeries, and medication.
Future medical costs for ongoing care or rehabilitation.
Therapy costs, including physical or mental health treatment.
Lost wages or reduced earning potential due to your injury.
Permanent disability, if applicable.
Pain and suffering caused by physical and emotional distress.
Emotional damages, such as anxiety or trauma.
Other compensatory and punitive damages.
An experienced Bard PowerPort injury attorney will work with you to calculate the full scope of your damages and build a case tailored to your circumstances. Contact Eric Cardenas LAW today to get started.
Eric Cardenas LAW: Bard PowerPort Injury Lawyers
With years of legal experience, Eric Cardenas LAW has helped secure millions of dollars in compensation for clients injured through no fault of their own.
If you or a loved one suffered harm due to a Bard PowerPort device, you may qualify to file a lawsuit against the manufacturer. Eric Cardenas LAW is committed to helping victims seek justice and holding negligent companies accountable.
Why Choose Eric Cardenas LAW?
Proven Results: Experience in securing compensation for clients.
Contingency Fee Basis: No upfront legal fees. You only pay if we win compensation for your case.
Free Consultations: Contact us to discuss your case at no cost or obligation.
Instant Eligibility Check: Use the Qualifying Quiz on this page to find out if you qualify for a Bard PowerPort lawsuit today.
If you’ve been injured by a defective medical device, we’re here to guide you through the legal process and fight for the justice you deserve. Reach out to us today.
Frequently Asked Questions
Has the Bard PowerPort Been Recalled?
Currently, there is no active recall for the Bard PowerPort device.
The FDA announced a recall in March 2020, but it was terminated in May 2022.
Despite the lack of an active recall, individuals who have experienced injuries related to the Bard PowerPort are encouraged to contact our experienced Bard PowerPort Injury Lawyers for a free consultation.
How Much Could a Bard PowerPort Settlement Be Worth?
The Bard PowerPort lawsuit is still in its early stages, and no settlements have been finalized.
However, estimates suggest potential settlements may range from $15,000 to over $300,000, depending on the severity of the injuries and the outcome of litigation.
These estimates are not guarantees but are projections based on past product liability cases. Contact Eric Cardenas LAW to discuss your eligibility and legal options.
What Is a Catheter Fracture?
A catheter fracture occurs when part of the catheter device breaks inside the patient’s body.
Fragments of silicone or polyurethane from the fractured catheter can enter the bloodstream, potentially causing severe complications as they travel through the circulatory system.
What Causes Catheter Fractures?
Catheter fractures often occur due to flex fatigue, which results from repeated bending or movement near the PowerPort. Over time, this wear can weaken the device, leading to fractures.
What Is Catheter Migration?
Catheter migration is a rare but serious complication where the catheter detaches from the PowerPort device and moves to other parts of the body.
Fragments of the catheter can travel to vital organs, such as the heart, where they may become lodged, requiring emergency surgery to remove.
What Causes a Catheter Migration?
Catheter migration can result from:
Improper installation of the PowerPort device.
Manufacturing defects, which compromise the device’s stability.
The manufacturer, Bard Access Systems, may be held liable if a manufacturing defect caused dislodgment and migration.
If you’ve experienced complications such as catheter migration, contact Eric Cardenas LAW for a free consultation to explore your legal options.


